✔ 100% Authentic Product
👁️ Currently Viewing 2479
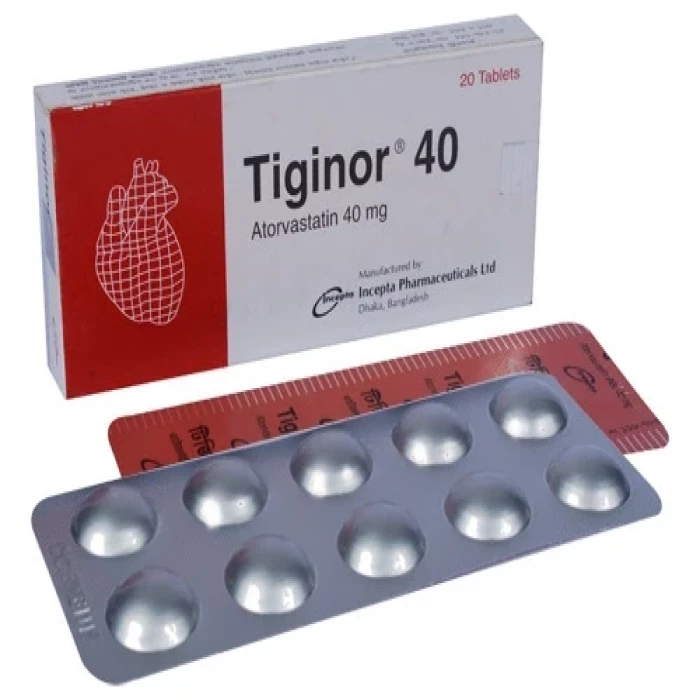
Tiginor 40 20Pcs (Box)
Tablet. Generic Name: Atorvastatin Calcium 10mg. Manufacturer: Incepta Pharmaceuticals Limited
Discount
Price: ৳ 523
MRP:
৳
550
5%
Off

100% Genuine Products, Guaranteed

Safe & Secure Payments, Always

Fast, Secure & Efficient Delivery
Proper Packaging
 Cash on Delivery - All over Bangladesh
Cash on Delivery - All over Bangladesh Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59
Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59 Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125
Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125
 ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে
ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে ফ্রি ডেলিভারিঃ - ৫৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
ফ্রি ডেলিভারিঃ - ৫৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
📲 মোবাইল অ্যাপ অর্ডারে সাশ্রয় বেশী
-
Google Play Store থেকে ডাউনলোড
-
Apple Store থেকে ডাউনলোড
100% Genuine Products, Guaranteed
Safe & Secure Payments, Always
Fast, Secure & Efficient Delivery
Proper Packaging
 Cash on Delivery - All over Bangladesh
Cash on Delivery - All over Bangladesh Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59
Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59 Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125
Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125 ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে
ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে ফ্রি ডেলিভারিঃ - ৫৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
ফ্রি ডেলিভারিঃ - ৫৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে- Google Play Store থেকে ডাউনলোড
- Apple Store থেকে ডাউনলোড
📲 মোবাইল অ্যাপ অর্ডারে সাশ্রয় বেশী
✅ Description:
Indications
Tiginor is indicated as an adjunct to diet to reduce elevated total cholesterol, LDL cholesterol, apolipoprotein B (Apo-B) and triglycerides levels in following diseases when response to diet and other non-pharmacological measures is inadequate.
To reduce total cholesterol and LDL cholesterol in patients with heterozygous and homozygous familial hypercholesterolaemia.
To reduce elevated cholesterol and triglycerides in patient with mixed dyslipidemia (Fredrickson Type Ia and Ib).
For the treatment of patients with elevated serum triglyceride levels in hypertriglyceridaemia (Fredrickson Type IV).
For the treatment of patients with dysbetalipoproteinaemia (Fredrickson Type III).
To reduce cardiac ischaemic events in patients with asymptomatic or mild to moderate symptomatic coronary artery disease with elevated LDL-cholesterol level.
To reduce total and LDL-cholesterol concentrations patients with hypercholesterolemia associated with or exacerbated by diabetes mellitus or renal transplantation.
Pharmacology
Atorvastatin is a selective inhibitor of HMG-CoA reductase. This enzyme is the rate-limiting enzyme responsible for the conversion of HMG-CoA to mevalonate, a precursor of sterols, including cholesterol. Atorvastatin lowers plasma cholesterol and lipoprotein levels by inhibiting HMG-CoA reductase and cholesterol synthesis in the liver and increases the number of hepatic LDL receptors on the cell surface for enhanced uptake and catabolism of LDL.
Absorption: Atorvastatin is rapidly absorbed after oral administration; maximum plasma concentrations occur within 1 to 2 hours. Extent of absorption increases in proportion to Atorvastatin dose. The absolute bioavailability of Atorvastatin (parent drug) is approximately 14% and the systemic availability of HMG-CoA reductase inhibitory activity is approximately 30%.
Distribution: Mean volume of distribution of Atorvastatin is approximately 381 liters. Atorvastatin is 98% bound to plasma proteins. A blood/plasma ratio of approximately 0.25 indicates poor drug penetration into red blood cells. Based on observations in rats, Atorvastatin is likely to be secreted in human milk.
Metabolism: Atorvastatin is extensively metabolized to ortho- and parahydroxylated derivatives and various beta-oxidation products. In vitro inhibition of HMG-CoA reductase by ortho- and parahydroxylated metabolites is equivalent to that of Atorvastatin. Approximately 70% of circulating inhibitory activity for HMG-CoA reductase is attributed to active metabolites. In vitro studies suggest the importance of Atorvastatin metabolism by cytochrome P450 3A4, consistent with increased plasma concentrations of Atorvastatin in humans following coadministration with erythromycin, a known inhibitor of this isozymes. In animals, the ortho-hydroxy metabolite undergoes further glucuronidation.
Excretion: Atorvastatin and its metabolites are eliminated primarily in bile following hepatic and/or extra-hepatic metabolism; however, the drug does not appear to undergo enterohepatic recirculation. Mean plasma elimination half-life of Atorvastatin in humans is approximately 14 hours, but the half-life of inhibitory activity for HMG-CoA reductase is 20 to 30 hours due to the contribution of active metabolites. Less than 2% of a dose of Atorvastatin is recovered in urine following oral administration.
Dosage & Administration
Primary hypercholesterolaemia and combined hyperlipidaemia-
Adults: Usually 10 mg once daily; if necessary, may be increased at intervals of at least 4 weeks to max. 80 mg once daily.
Child (10-18 years): Initially 10 mg once daily, increased if necessary at intervals of at least 4 weeks to usual max. 20 mg once daily.
Familial hypercholesterolaemia-
Adults: Initially 10 mg daily, increased at intervals of at least 4 weeks to 40 mg once daily; if necessary, further increased to max. 80 mg once daily (or 40 mg once daily combined with anion-exchange resin in heterozygous familial hypercholesterolaemia).
Child (10-18 years): Initially 10 mg once daily, increased if necessary at intervals of at least 4 weeks to usual max. 80 mg once daily.
Prevention of cardiovascular events-
Adults: Initially 10 mg once daily adjusted according to response.
Another guideline:
The patient should be placed on a standard cholesterol-lowering diet before receiving Atorvastatin and should continue on this diet during treatment with Atorvastatin. The recommended starting dose of Atorvastatin is 10 or 20 mg once daily. Patients who require a large reduction in LDL-C (more than 45%) may be started at 40 mg once daily. The dosage range of Atorvastatin is 10 to 80 mg once daily. Atorvastatin can be administered as a single dose at any time of the day, with or without food. After initiation of Atorvastatin lipid levels should be analyzed within 2 to 4 weeks and dosage adjusted accordingly.
In hypercholesterolemia in Pediatric Patients (10-17 years of age) the recommended starting dose of Atorvastatin is 10 mg/day; the maximum recommended dose is 20 mg/day (doses greater than 20 mg have not been studied in this patient population). Doses should be individualized according to the recommended goal of therapy. Adjustments should be made at intervals of 4 weeks or more.
* চিকিৎসকের পরামর্শ মোতাবেক ঔষধ সেবন করুন'
Interaction
The risk of myopathy during treatment with drugs of this class is increased with concurrent administration of cyclosporine, fibric acid derivatives, niacin (nicotinic acid), erythromycin, azole antifungals.
Antacid: When atorvastatin and antacid suspension were coadministered, plasma concentrations of atorvastatin decreased approximately 35%. However, LDL-C reduction was not altered.
Colestipol: Plasma concentrations of atorvastatin decreased approximately 25% when colestipol and atorvastatin were coadministered. However, LDL-C reduction was greater when atorvastatin and colestipol were coadministered than when either drug was given alone.
Digoxin: When multiple doses of atorvastatin and digoxin were coadministered, steady-state plasma digoxin concentrations increased by approximately 20%. Patients taking digoxin should be monitored appropriately.
Erythromycin: In healthy individuals, plasma concentrations of atorvastatin increased approximately 40% with coadministration of atorvastatin and erythromycin, a known inhibitor of cytochrome P450 3A4.
Oral Contraceptives: Coadministration of atorvastatin and an oral contraceptive increased AUC values for norethindrone and ethinyl estradiol by approximately 30% and 20%. These increases should be considered when selecting an oral contraceptive for a woman taking atorvastatin.
Warfarin: Tiginor had no clinically significant effect on prothrombin time when administered to patients receiving chronic warfarin treatment.
Contraindications
Atorvastatin should not be used in patient with hypersensitivity to any component of this medication. Atorvastatin is contraindicated in active liver disease or unexplained persistent elevations of serum transaminases. It is also contraindicated in patient with history of serious adverse reaction to prior administration of HMG-CoA reductase inhibitors.
Side Effects
Tiginor is generally well-tolerated. The most frequent side effects related to Tiginor are constipation, flatulence, dyspepsia, abdominal pain. Other side effects includes infection, headache, back pain, rash, asthenia, arthralgia, myalgia.
Pregnancy & Lactation
Pregnancy: Atorvastatin is contraindicated during pregnancy. Safety in pregnant women has not been established. No controlled clinical trials with atorvastatin have been conducted in pregnant women. Rare reports of congenital anomalies following intrauterine exposure to HMG-CoA reductase inhibitors have been received. Animal studies have shown toxicity to reproduction. Maternal treatment with atorvastatin may reduce the fetal levels of mevalonate which is a precursor of cholesterol biosynthesis. Atorvastatin should not be used in women who are pregnant, trying to become pregnant or suspect they are pregnant. Treatment with atorvastatin should be suspended for the duration of pregnancy or until it has been determined that the woman is not pregnant
Lactation: It is not known whether atorvastatin or its metabolites are excreted in human milk. In rats, plasma concentrations of atorvastatin and its active metabolites are similar to those in milk. Because of the potential for serious adverse reactions, women taking atorvastatin should not breastfeed their infants. Atorvastatin is contraindicated during breastfeeding.
Precautions & Warnings
Liver effects: Liver function tests should be performed before the initiation of treatment and periodically thereafter. Tiginor should be used with caution in patients who consume substantial quantities of alcohol or have a history of liver disease. Tiginor therapy should be discontinued if markedly elevated CPK levels occur or myopathy is diagnosed or suspected.
Use in Special Populations
Geriatric: Plasma concentrations of Tiginor are higher (approximately 40% for Cmax and 30% for AUC) in healthy elderly subjects (age 65 years) than in young adults. Clinical data suggest a greater degree of LDL-lowering at any dose of drug in the elderly patient population compared to younger adults.
Pediatric: Pharmacokinetic data in the pediatric population are not available.
Gender: There is no clinically significant difference in LDL-C reduction with Tiginor between men and women.
Renal Insufficiency: Renal disease has no influence on the plasma concentrations or LDL-C reduction of Tiginor; thus, dose adjustment in patients with renal dysfunction is not necessary.
Hemodialysis: Hemodialysis is not expected to significantly enhance clearance of Tiginor since the drug is extensively bound to plasma proteins.
Hepatic Insufficiency: In patients with chronic alcoholic liver disease, plasma concentrations of Tiginor are markedly increased.
Overdose Effects
Specific treatment is not available for atorvastatin overdose. The patient should be treated symptomatically and supportive measures instituted, as required. Liver function tests should be performed and serum CK levels should be monitored. Due to extensive atorvastatin binding to plasma proteins, hemodialysis is not expected to significantly enhance atorvastatin clearance.
Therapeutic Class
Other Anti-anginal & Anti-ischaemic drugs, Statins
Storage Conditions
Keep in a dry place away from light and heat. Keep out of the reach of children.
⚠️Disclaimer:
At ePharma, we’re committed to providing accurate and accessible health information. However, all content is intended for informational purposes only and should not replace medical advice from a qualified physician. Please consult your healthcare provider for personalized guidance. We aim to support, not substitute, the doctor-patient relationship.